ImagineIT Phase 3: Action Plan
Identify Desired Results
The big idea and major focus in my sophomore chemistry class this year will be to give students opportunities and tools to connect chemistry to their daily lives and to approach problems with a scientific approach. In the past, students have left the class at the end of the year with a feeling of accomplishment for getting through one of the tougher courses in the school due to its abstract nature. However, the science department is well aware that many of our students, freshmen up to seniors, do not retain more than a few pieces of knowledge from each course and possibly a skill or two in a lab or engineering context. We see this each year in the confused looks we get from a new set of students when we ask, “Remember when you learned this last year?”
The key to making these ideas, concepts, and skills stick is to anchor them in something students already know (Heath & Heath, 2010). This can be tricky at a school that pulls its students from middle schools across the city since their prior knowledge will vary greatly; however, many of them have very similar daily experiences that we can tap into to make these connections. Everything around us can be connected to chemistry in some way and the chemistry can explain natural phenomena and “mysteries” that students witness in their lives. These mysteries bring about a curiosity that makes us want to fill this “gap” in our knowledge. As explained by Heath and Heath, “one important implication of the “gap theory” is that we need to open gaps before we close them. Our tendency is to tell students the facts. First, though, they must realize they need them” (2010).
By creating a comfortable environment which allows for misconceptions to be brought out into the open, I can begin to work toward the true purpose for this year. We will start to share our daily experiences and the questions that our natural curiosity generates; there are no “silly” questions. As we slowly rebuild our knowledge base that has strong foundations in what we already know, students should be able to examine and attack novel situations that span across multiple contexts, just like we see in our ever-changing, interconnected world. They should leave with the skills needed to “meet the present and the future in informed ways” (Mansilla & Gardner, 2008).
Determine Acceptable Evidence
Assessing whether students are making connections between the chemistry and their world around them requires students to bring their thinking out of the privacy of their own mind. “Performances of understanding require students to show their understanding in an observable way. They make students’ thinking visible” (Wiggins & McTighe, 2005). A great way to do this is through both journaling and sharing of questions and ideas. Keeping a journal and posing questions that require students to think about their struggles with understanding will help to create an open learning environment where students feel comfortable admitting they do not fully grasp something. Though this journal will primarily be between me and the individual student, it is a safe place to start to encourage a comfortable classroom.
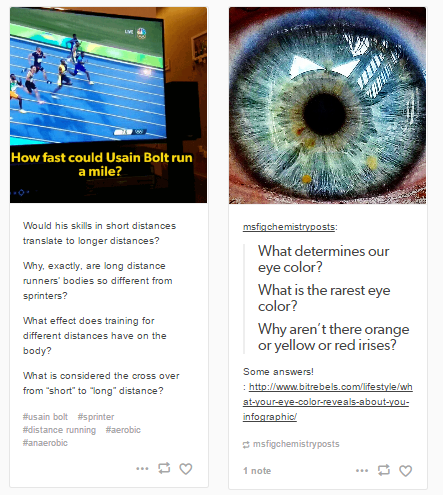
A step up from the journal will be a forum where students will be required to share at least one picture or video of something, per semester, that they experienced that made them wonder. A Tumblr page has been created where students can edit and submit photos with a brief explanations of what they saw and why they think this may have happened. The goal is not that they necessarily get the explanation correct, but that they are now viewing the world through a more inquisitive lens. This idea will be made clear to students so that they do not feel pressure to force something because they are unsure of how to explain it. These posts can also give me insight on what my students see and are interested in on a daily basis and can help guide explanations that directly connect to these experiences.
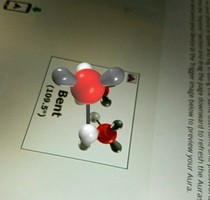
Performances of understanding require students to use what they know in a new situation. One clear example of this will take place after students spend time learning about how atoms come together to form molecules and what the shape of the molecule tells us about its properties. They will start by exploring simple molecules, such as water, methane, and ammonia, and then begin to choose more complex molecules to re-apply this knowledge to such as Vitamin C, caffeine, or teflon. This helps them to transfer their knowledge to a new situation and see how the pieces fit and apply in a similar way. The properties of the complex molecule will match up with their experience with it (since they will choose a molecule that they are interested in and possibly have had some experience with), and they will need to clearly make this connection with a short anecdote.
Finally, to assess students on their ability to connect the chemistry explanation with their real world observations, they will be given another molecule and a few of its properties and asked to analyze its shape, how the properties are explained by this, and predict some other properties that the molecule may have (soluble in water, high melting point, etc.). “With deliberate and explicit instruction in how to transfer (and assessments that constantly demand such transfer), the learner must take what were initially bits of knowledge with no clear structure...and come to see them as part of a...more meaningful, more useful system” (Wiggins & McTighe, 2005).
Learning Experience and Instruction
Identify Desired Results
The big idea and major focus in my sophomore chemistry class this year will be to give students opportunities and tools to connect chemistry to their daily lives and to approach problems with a scientific approach. In the past, students have left the class at the end of the year with a feeling of accomplishment for getting through one of the tougher courses in the school due to its abstract nature. However, the science department is well aware that many of our students, freshmen up to seniors, do not retain more than a few pieces of knowledge from each course and possibly a skill or two in a lab or engineering context. We see this each year in the confused looks we get from a new set of students when we ask, “Remember when you learned this last year?”
The key to making these ideas, concepts, and skills stick is to anchor them in something students already know (Heath & Heath, 2010). This can be tricky at a school that pulls its students from middle schools across the city since their prior knowledge will vary greatly; however, many of them have very similar daily experiences that we can tap into to make these connections. Everything around us can be connected to chemistry in some way and the chemistry can explain natural phenomena and “mysteries” that students witness in their lives. These mysteries bring about a curiosity that makes us want to fill this “gap” in our knowledge. As explained by Heath and Heath, “one important implication of the “gap theory” is that we need to open gaps before we close them. Our tendency is to tell students the facts. First, though, they must realize they need them” (2010).
By creating a comfortable environment which allows for misconceptions to be brought out into the open, I can begin to work toward the true purpose for this year. We will start to share our daily experiences and the questions that our natural curiosity generates; there are no “silly” questions. As we slowly rebuild our knowledge base that has strong foundations in what we already know, students should be able to examine and attack novel situations that span across multiple contexts, just like we see in our ever-changing, interconnected world. They should leave with the skills needed to “meet the present and the future in informed ways” (Mansilla & Gardner, 2008).
Determine Acceptable Evidence
Assessing whether students are making connections between the chemistry and their world around them requires students to bring their thinking out of the privacy of their own mind. “Performances of understanding require students to show their understanding in an observable way. They make students’ thinking visible” (Wiggins & McTighe, 2005). A great way to do this is through both journaling and sharing of questions and ideas. Keeping a journal and posing questions that require students to think about their struggles with understanding will help to create an open learning environment where students feel comfortable admitting they do not fully grasp something. Though this journal will primarily be between me and the individual student, it is a safe place to start to encourage a comfortable classroom.
A step up from the journal will be a forum where students will be required to share at least one picture or video of something, per semester, that they experienced that made them wonder. A Tumblr page has been created where students can edit and submit photos with a brief explanations of what they saw and why they think this may have happened. The goal is not that they necessarily get the explanation correct, but that they are now viewing the world through a more inquisitive lens. This idea will be made clear to students so that they do not feel pressure to force something because they are unsure of how to explain it. These posts can also give me insight on what my students see and are interested in on a daily basis and can help guide explanations that directly connect to these experiences.
Performances of understanding require students to use what they know in a new situation. One clear example of this will take place after students spend time learning about how atoms come together to form molecules and what the shape of the molecule tells us about its properties. They will start by exploring simple molecules, such as water, methane, and ammonia, and then begin to choose more complex molecules to re-apply this knowledge to such as Vitamin C, caffeine, or teflon. This helps them to transfer their knowledge to a new situation and see how the pieces fit and apply in a similar way. The properties of the complex molecule will match up with their experience with it (since they will choose a molecule that they are interested in and possibly have had some experience with), and they will need to clearly make this connection with a short anecdote.
Finally, to assess students on their ability to connect the chemistry explanation with their real world observations, they will be given another molecule and a few of its properties and asked to analyze its shape, how the properties are explained by this, and predict some other properties that the molecule may have (soluble in water, high melting point, etc.). “With deliberate and explicit instruction in how to transfer (and assessments that constantly demand such transfer), the learner must take what were initially bits of knowledge with no clear structure...and come to see them as part of a...more meaningful, more useful system” (Wiggins & McTighe, 2005).
Learning Experience and Instruction
|
Context: The context of my ImagineIT project is a 10th grade chemistry class at Walter Payton College Prep HS. This is a selective enrollment public high school which means most students must earn specific grades in middle school and top scores on placement tests in order to be admitted. Because of this, we have some of the top students from every neighborhood in the city of Chicago, creating a very diverse student body and learning environment. Students’ prior knowledge in science is vastly different based on the middle school they came from. These students are also typically excellent test-takers, as this is one of the main reasons that they were accepted into a selective enrollment school. They know their way around tricky test strategies employed by teachers, but their weakness seems to display itself when students are given an open ended problem that requires non-linear thinking and the application of various disciplines and skills.
|
At this time, I plan on implementing this project in two chemistry classes that I am responsible for throughout the entire school year. We meet 2-3 times per week for a 1.5 hour block period in a chemistry lab/classroom. Nearly all of my students have a smartphone and/or access to the internet at home (I always conduct a survey at the beginning of the year to identify students that do not have access). Computer carts are available in our school, but the computers are typically slow and burdensome. I have a document camera and a standard projector in my room which can be connected to the camera on my phone. We also utilize our class website which is run through Moodle. There are options to upload documents, pictures, and videos from both the teacher and the students. Students are able to post to a forum that is seen by everyone in the class and they can also post to a journal that is only visible to the teacher.
Content: The primary goal for my class this year is to make chemistry accessible and engaging to all students by connecting it to their everyday lives. I want them to begin to see the world through a new lens, where they question discrepant events and phenomena and actually have some of the tools and knowledge to explain these events. Some of the biggest issues we face in the science department is dispelling misconceptions and getting students to actually retain big ideas year after year.
In chemistry, problems arise when we zoom all the way in to a scale that is not visible to the human eye. You can ask a student what their chair is made up of on the most basic level and they can tell you atoms, protons, neutrons, and so on, but ask them if they actually believe that fact and you may be surprised (or not)! Then, we throw a lot of math on top of these abstract concepts and it becomes difficult for students to see what their solution actually means, let alone if it is correct. It is my hope to continually “zoom out” as often as we “zoom in” in order to constantly base this new information in experiences that my students are familiar with, and therefore, make it stick.
Pedagogy: Since my goals focus on allowing students to uncover content through real world application, a project-based learning (PBL) approach aligns perfectly to help me achieve this. “In a PBL classroom, students learn in the context of a problem to be solved. The responsibility for learning is with the students, not with the facilitator” (Ram, 1999). By creating or choosing a problem with student interest in mind, we can work backwards to move forward. In other words, we typically start with the most basic concepts, build on those, then finish a unit with a project that brings it all together. PBL flips this process while also allowing students to discover the content as they need it for the overarching problem/project. This gives students a “need to know” and lets them anchor the newly obtained knowledge in a place that has meaning (Heath & Heath, 2010), attacking the problem of amnesia where students regularly forget what they have learned (Shulman, 1999).
Content: The primary goal for my class this year is to make chemistry accessible and engaging to all students by connecting it to their everyday lives. I want them to begin to see the world through a new lens, where they question discrepant events and phenomena and actually have some of the tools and knowledge to explain these events. Some of the biggest issues we face in the science department is dispelling misconceptions and getting students to actually retain big ideas year after year.
In chemistry, problems arise when we zoom all the way in to a scale that is not visible to the human eye. You can ask a student what their chair is made up of on the most basic level and they can tell you atoms, protons, neutrons, and so on, but ask them if they actually believe that fact and you may be surprised (or not)! Then, we throw a lot of math on top of these abstract concepts and it becomes difficult for students to see what their solution actually means, let alone if it is correct. It is my hope to continually “zoom out” as often as we “zoom in” in order to constantly base this new information in experiences that my students are familiar with, and therefore, make it stick.
Pedagogy: Since my goals focus on allowing students to uncover content through real world application, a project-based learning (PBL) approach aligns perfectly to help me achieve this. “In a PBL classroom, students learn in the context of a problem to be solved. The responsibility for learning is with the students, not with the facilitator” (Ram, 1999). By creating or choosing a problem with student interest in mind, we can work backwards to move forward. In other words, we typically start with the most basic concepts, build on those, then finish a unit with a project that brings it all together. PBL flips this process while also allowing students to discover the content as they need it for the overarching problem/project. This gives students a “need to know” and lets them anchor the newly obtained knowledge in a place that has meaning (Heath & Heath, 2010), attacking the problem of amnesia where students regularly forget what they have learned (Shulman, 1999).
|
A second instructional strategy that works at fixing the same problems of retaining information and breaking down misconceptions is reflecting through journaling (private) and sharing daily experiences (public). Journaling will occur throughout every phase of a unit to capture preconceptions at the beginning of the unit, struggles within the unit, and reflections at the end of a unit. Sharing daily experiences can also happen at any point in time but will be primarily tracked through the use of a class Tumblr. Students will be able to share daily experiences from any facet of their lives and begin to question what they witness. This encourages them to think more like a scientist and pick apart these phenomena so that they can start making connections between chemistry and their surroundings.
|
|
Technology: A project-based assignment can require a variety of technology depending on the project. Smartphones and laptops are primarily the tool of choice since they can be used for research, simulations, videos, and journaling. Moodle is a site that our school uses for class websites and it is something that both parents and students are very familiar with. Because of this, I plan to keep a “home base” within our moodle page that can house links to external resources. Moodle has a journal option where students can submit journal entries directly to the teacher, and they can also include pictures, videos, and hyperlinks. There are many other journaling programs out there but I believe that the simplicity of the Moodle Journal will be sufficient for its purpose. A few ways to bring abstract, microscopic concepts to life are through simulations, models, and augmented reality. The University of Colorado Boulder has created a series of interactive simulations (PhET) which “provide dynamic access to multiple representations, make the invisible visible, scaffold inquiry, and allow for safe and quick access to multiple trials” (Moore, et. al., 2014). These are fully interactive, meaning the students can raise and lower the temperature, increase concentration of reagents, decrease the volume, and change many other factors in order to see the effect on substance being tested. Aurasma is another program that allows students to create and manipulate augmented reality right on their phones. Although full manipulation of the three dimensional model is still not possible, this tool fills a gap that the PhET simulations cannot. Students can rotate the model, a molecule for example, in three dimensions to fully understand how it exists in space and therefore, how it interacts with other substances. |
References
Heath, C., & Heath, D. (2010). Teaching that Sticks. 1-11.
Mansilla, V.B., & Gardner, H. (2008). Disciplining the mind. Educational Leadership. 65(5), 14-19.
Moore, E. B., Chamberlain, J. M., Parson, R., & Perkins, K. K. (2014). PhET Interactive Simulations: Transformative Tools for Teaching Chemistry, Journal of Chemical Education, 91(8), 1191-1197.
Ram, P. (1999). Problem-based learning in undergraduate education. Journal of Chemical Education, 76(8), 1122-1126.
Shulman, L.S. (1999). What is learning and what does it look like when it doesn’t go well? Change, 31(4), 10-17.
Wiggins, G., & McTighe, J. Understanding understanding. In Understanding by Design, expanded 2nd Edition, ASCD